Haem.io
Intelligent Diagnostics for Precision Haematology
Haem.io
Intelligent Diagnostics for Precision Haematology
The Problem
Haematology diagnosis is broken
Unstructured Data
Clinicians manually synthesize genetic markers from cytogenetics, flow cytometry, and molecular data — each in different formats with no standardization.
Overwhelmed Diagnostician
Hours spent manually extracting information before any diagnostic logic can begin. Smaller trusts outsource to 5 specialist centres at great cost.
Impossible Complexity
WHO 2022 and ICC 2022 guidelines contain hundreds of interconnected diagnostic pathways — too complex for consistent human application.
Early Traction
Product built, clinical partnerships secured, international validation
Working Product
AML classifier, MDS classifier, and clinical trial matching — all LIVE in production. Covers all WHO 2022 and ICC 2022 subtypes.
Pharma Grant Pipeline
Pfizer, Servier, Jazz, and J&J in active discussions for ~£210k in grant funding. All expressing strong interest.
£7M National Grant
Co-applicant on £7M UK AML Research Network grant for national AML relapse MDT standardisation.
4 NHS Endorsements
Letters of support from consultant haematologists at The Christie, Royal Devon, Blackpool Teaching Hospitals, and the UK AML Research Network.
International Interest
Cambodia demo completed — strong interest in deployment. Built on WHO/ICC global standards — works in any country without modification.
IP Strategy
IP assignment from Manchester Foundation Trust in active negotiation. Core codebase independently developed by founding team.
The Solution
End-to-end AI-powered haematology diagnostics
Haem.io is the only platform that combines AI data extraction with formal diagnostic logic engines to deliver complete haematology classification, risk stratification, and treatment guidance.
Step 1: AI Extracts Structure from Chaos
Unstructured lab reports → clean, structured data in seconds
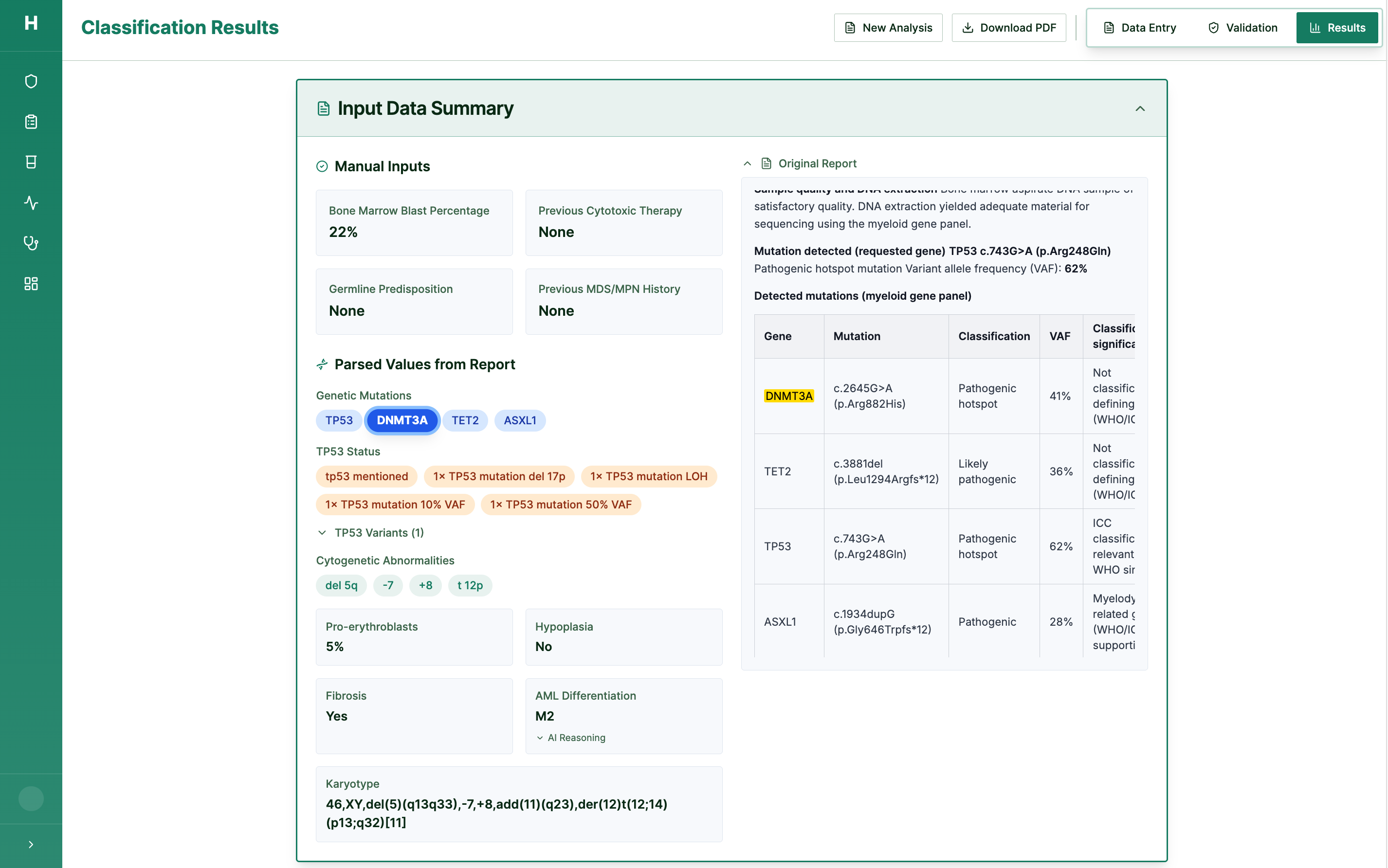
Step 2: Logic Engine Runs Diagnostic Pathways
WHO 2022 & ICC 2022 classification — fully explainable, fully auditable
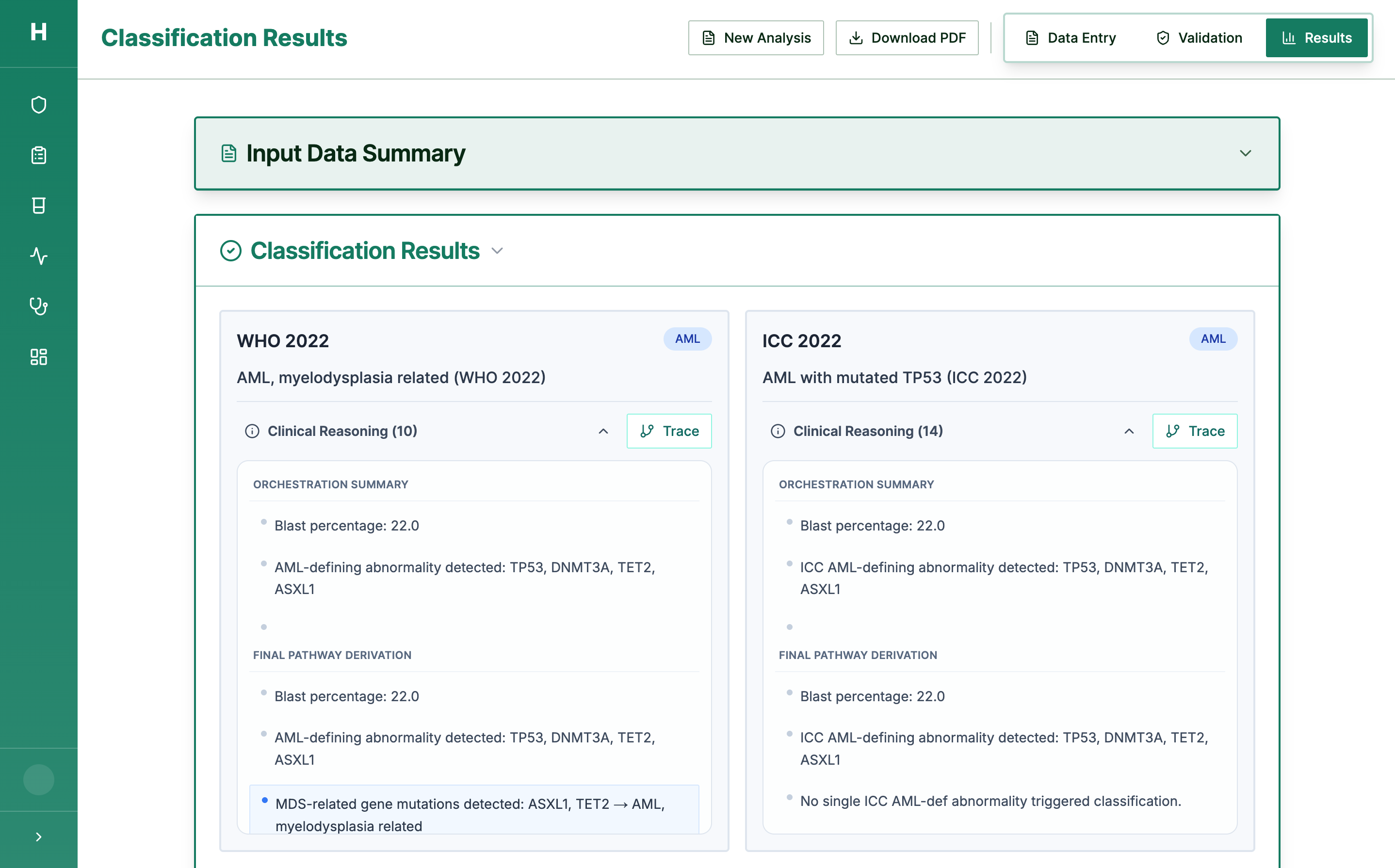
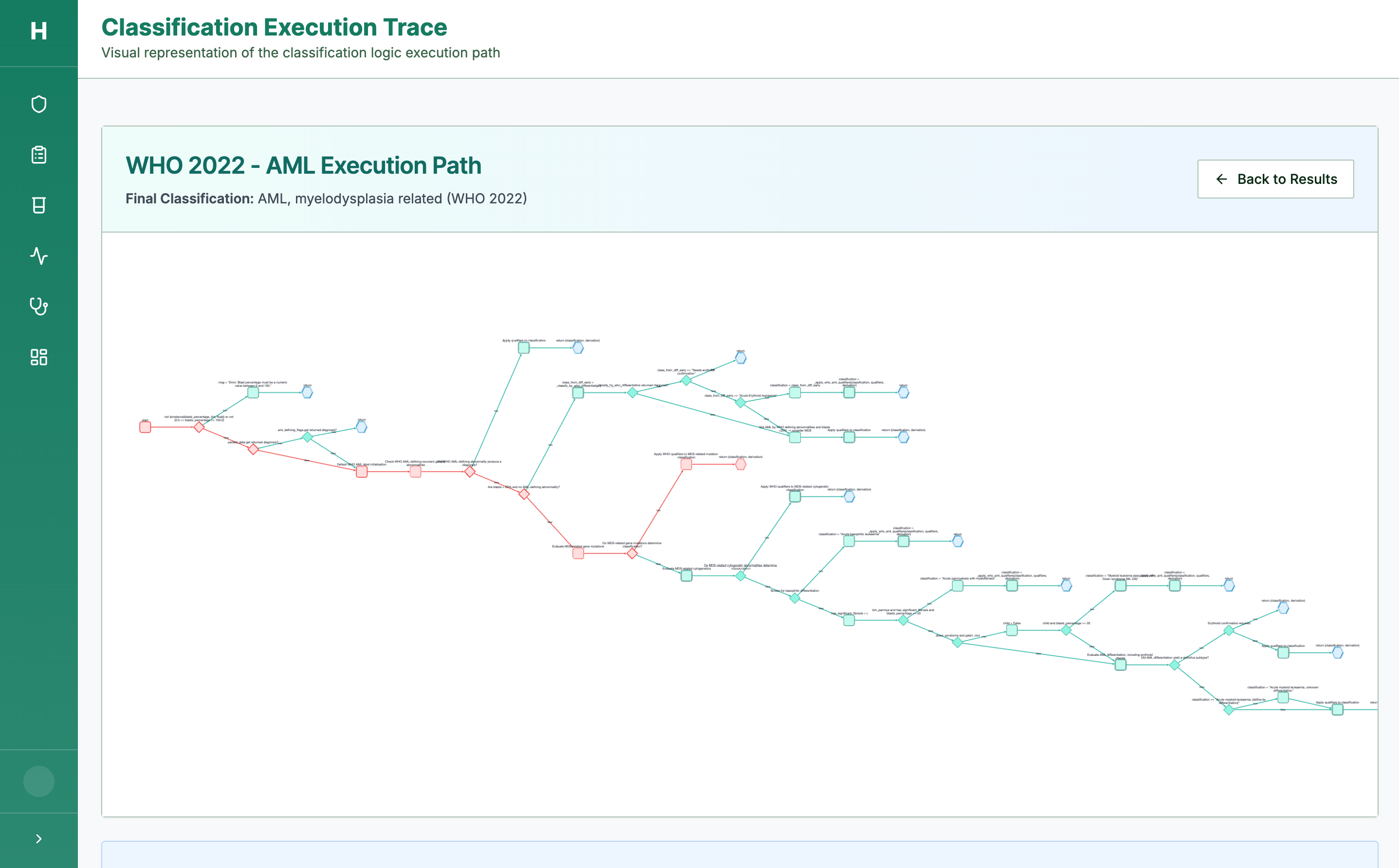
Step 3: Every Decision is Traceable
Full execution trace — every criterion checked, every pathway followed
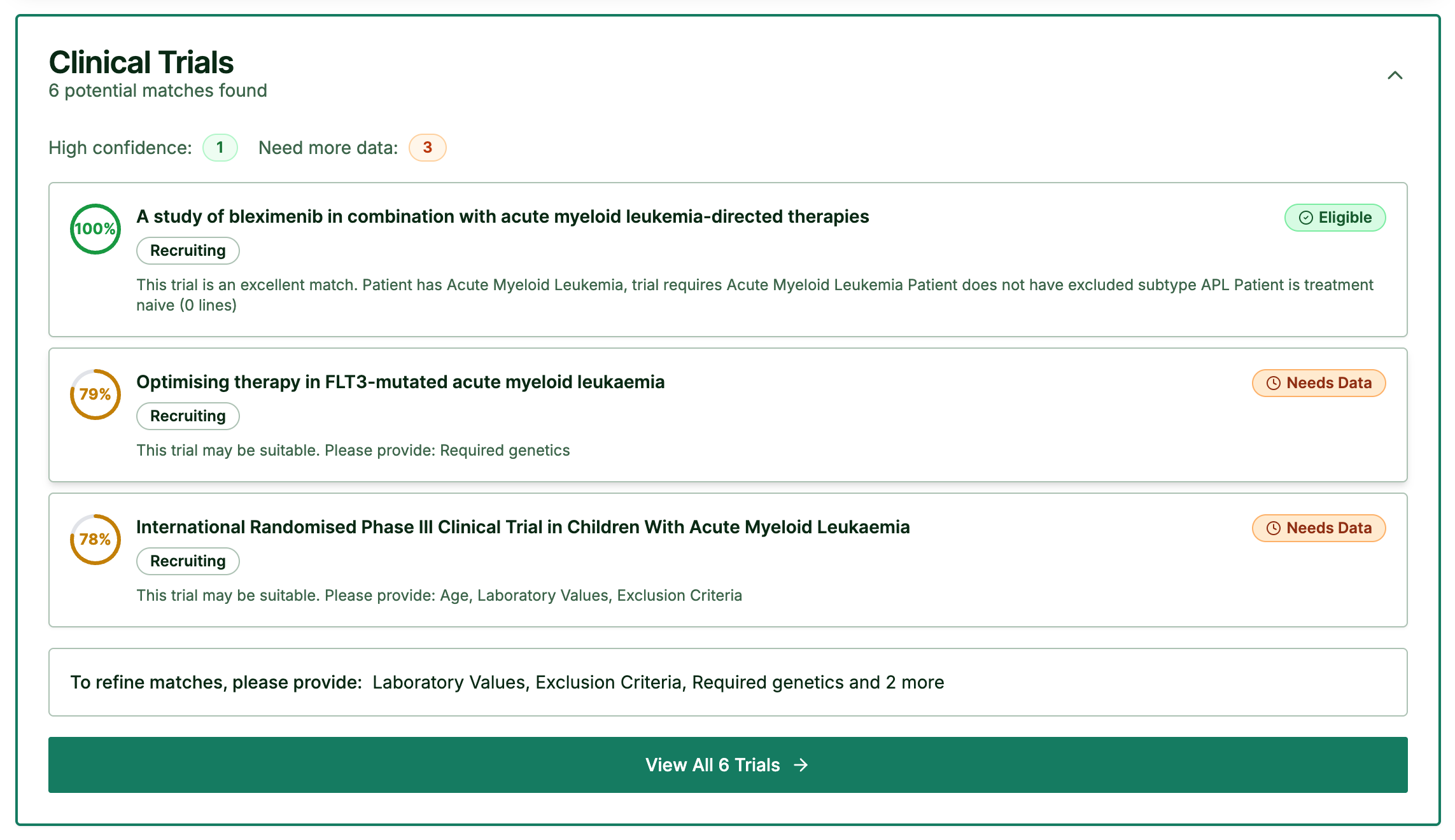
Step 4: Clinical Trial Matching
Automatically matches patients to eligible trials based on their diagnosis
How It Works
Not a black box: transparent, explainable, auditable
flow cytometry CD34+, CD117+, HLA-DR+...
molecular NPM1 mutation detected..."
WBC: 45.3
Blasts: 72%
Diagnostic
Pathways
criteria met, pathways followed
Acute Myeloid Leukemia
NPM1-mutated
Why We Win
What makes us hard to replicate
Zero Integration Friction
Works with any report format — PDF, scanned documents, text files. No IT integration required. Hospitals start using it immediately without changing existing systems.
Clinical Expertise as Code
Thousands of hours of consultant haematologists iterating directly with engineers to codify WHO/ICC diagnostic logic. This clinical-engineering feedback loop took years — it cannot be replicated by bolting AI onto existing platforms.
Full Diagnostic Pipeline
Not just a diagnosis — we deliver treatment recommendations, clinical trial matching, risk stratification, and MRD monitoring. A complete decision support system, not a point solution.
Explainable by Design
Our logic engine produces a full derivation trace — every diagnostic decision is auditable. Critical for clinical trust, regulatory approval, and clinician adoption.
Global by Default
Built on WHO 2022 and ICC 2022 international standards. Works in any country, any hospital, from day one. No localization or adaptation needed — the diagnostic logic is universal.
Competitive Landscape
We are the only end-to-end solution for modern haematology
Lab Automation
e.g., Scopio Labs
AI-powered morphology analysis
Gap: No genomic integration or WHO/ICC classification
Genomics Platforms
e.g., SOPHiA GENETICS
Genomic sequencing analysis
Gap: No automated diagnosis or risk stratification
AI Pathology
e.g., PathAI
Solid tumor histology
Gap: Not built for haematology complexity
Our Unique Position
First to fuse morphology, flow cytometry, and genomics into a single diagnostic workflow
Purpose-built for WHO & ICC 2022 mutation-based criteria — not retrofitting old systems
Beyond classification — risk stratification (ELN 2022, IPSS-M), treatment guidance, trial matching
Why can't well-funded competitors build this? Classification logic required thousands of hours of specialist haematologists iterating directly with engineers. It's codified clinical judgement built over years — not a dataset you can license or an algorithm you can train.
Market Opportunity
All cancer diagnostics are going genomic. Haematology is the leading edge.
Solve the hardest cancer first — the framework, credibility, and regulatory pathway extend to every cancer type as genomic classification becomes standard.
Business Model
SaaS licensing to NHS trusts, private hospitals, and diagnostic labs
Annual SaaS Licensing
NHS Trusts
£50k-£100k/year
Private Hospitals
£75k-£150k/year
Diagnostic Labs
£100k-£200k/year
Year 1 (2026)
Pilots & clinical validation with 4 NHS trusts. No revenue.
Year 2 (2027)
Class I registration complete. First 2-3 contracts signed Q2-Q3.
Year 3 (2028)
12 customers. ~£700k revenue. Breakeven.
ROI: Why Hospitals Switch
Outsourcing costs thousands per case. Haem.io costs a fraction.
~140+ NHS trusts lack specialist haematology diagnostics and outsource at significant cost.
A trust processing 50 cases/year at £2k = £100k. Haem.io license: £50-100k. The product pays for itself.
~40,000 AML/MDS cases diagnosed in the UK annually
Clinical Validation
Endorsed by leading NHS haematologists
Expansion Path
Starting with the hardest problem first
AML + MDS
The most complex blood cancers — hundreds of subtypes requiring integration of cytogenetics, flow cytometry, molecular data, and clinical history
Other Blood Cancers
Lymphomas, multiple myeloma, and other haematological malignancies
Solid Tumors
Expanding our AI + logic framework to genomics-driven solid tumor diagnostics
International interest: Cambodia demo completed with strong reception. WHO/ICC global standards = no localization needed.
The Team
Clinical expertise meets technical depth

Robert Lee
CEO & Co-Founder
BSc Computer Science, Manchester. Cancer survivor driving the mission. Leaving senior role at Coinbase (>70% pay cut) to build Haem.io full-time. Background in FinTech at LSEG and FlexTrade.

Dr. Daniel Clarke
CTO & Co-Founder
PhD Physics, University of Manchester & CERN. Former UK Civil Service data scientist. Leading platform architecture and AI strategy.

Dr. John Burthem
Chief Medical Officer & Co-Founder
FRCP, FRCPath. Lead Haematology Diagnostician at Manchester Foundation Trust. 50+ peer-reviewed publications. Co-inventor of Haem.io's clinical logic.

Dr. Luke Carter-Brzezinski
Clinical Director & Co-Founder
FRCPath. Consultant Haematologist at MFT. Leading clinical outreach, pilot studies, and validation strategy across the NHS.
All founders taking significantly below-market salaries. Next hire: Commercial/Sales Lead to drive NHS adoption post-registration.
The Ask
£750k seed round. 18 months to Series A.
Seed Investment | 18-Month Runway
Team (18 months)
Regulatory & Pilots
Infrastructure & Operations
Non-dilutive funding pipeline: ~£210k in pharma grant discussions (Pfizer, Servier, Jazz, J&J). Co-applicant on £7M UK AML Research Network grant.
Haem.io
Precision diagnostics for every haematologist, everywhere.
Built by clinicians. Powered by AI. Explainable by design.